
New information is coming in about the advancement of HIV prevention.
Scientists and researchers have been working on an injectable for HIV prevention since back in 2017. Now three years later, they have study results to show how progress on the prospective shot is going. And we’re happy to hear that it’s going well.
According to Reuters, the drug company GlaxoSmithKline has seen promising results form its current drug trial. The company says that taking injections of its cabotegravir drug once a month is more effective than Gilead’s daily Truvada pill. Specifically, it was found to be 69% more effective than Truvada.
Kimberly Smith, the head of research for GlaxoSmithKline’s ViiV unit, noted how the injection could greatly change the quality of life factor for men who have sex with other men.
“Individuals have to show up every eight weeks in the clinic for the injection but in-between there is not a need to take a pill daily, so you really change the equation for adherence with a long acting (drug),” Smith explained.
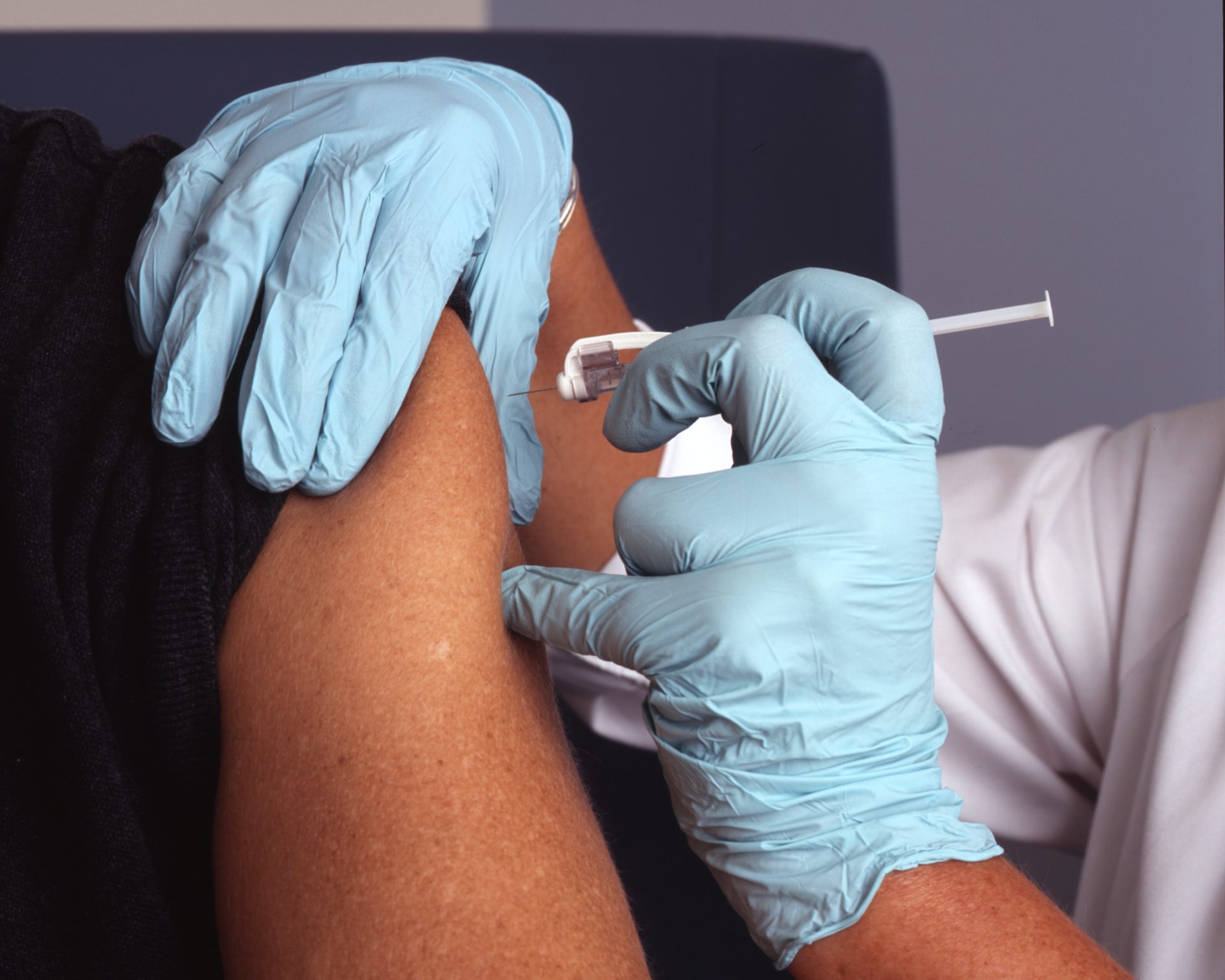
Now that the drug trial has shown such great results, GSK will speak to drug regulators about a possible approval of their drug. Though, the company has already won approval in Canada for cabotegravir as one of the two key ingredients in the long-acting HIV treatment combination Cabenuva.
This is just the start of a new age of HIV prevention and treatment. Currently, the drug company Gilead is leading in the HIV treatment market. Just last year, the company announced a new and more effective drug. This new daily pill was shown to be less toxic to the kidneys and bones. Plus, it is more effective in preventing HIV. But, it will also be more expensive than Truvada. Though, cheaper generic versions of Truvada are coming to the United States this September. This is due to Gilead’s patent expiring and other companies being able to create their own versions of the drug.
With all of that in mind, it seems the world of HIV prevention and treatment is about to get more competitive. But for us patients, that means we’ll have more options in how we protect/treat ourselves. And that’s what’s most important.
Source: Reuters, Science Mag

Even though the patent expires for Truvada, the price will not drop dramatically because of some under-handed manipulation on the part of Gilead. They sold the patent to a company in another country, with residuals, and so the patent will not really expire.
que contraindicaciones trae y cual es la formula quimica de esta vacuna prevetiva contra el vih y por que se debe tomar cada mes no es mas facil una al año… digo yo no se .. y se puede tener relaciones sexuales sin condon evitando al 100 % el vih sin olvidar que se puede contagiar otras ets…